IJMS Editor’s Choice – the metabolic cost of calcification
As a group, oysters are the marine animals most raised in aquaculture. While there has been continued growth in global oyster production over the past decades, recent failures of larval Pacific oyster hatchery production along the U.S. West Coast highlight negative impacts of ocean acidification on marine aquaculture. As well as its importance as a source of seafood, the Pacific oyster is emerging as a model marine organism for study of development, physiology, and genetics. Responses to ocean acidification can involve high metabolic costs. In this study, the authors quantify the effect of ocean acidification on the allocation of metabolic energy and the cost of shell-formation during early larval development.
Within the first two days of development, the Pacific oyster grows a shell that is six times heavier than the mass of its whole body. Accomplishing this high rate of shell growth requires up to two-thirds of total metabolic energy. Yet, surprisingly, regardless of exposure to experimental ocean acidification, the amount of metabolic energy required and the maternally-endowed energy reserves utilized to complete development is the same. Consequently, decreased calcification rates resulting in smaller shells that are commonly observed under experimental ocean acidification are not caused by a limitation of biochemical energy reserves.
Despite the substantial impact of experimental ocean acidification on shell formation, different families of Pacific oyster range in their ability to make shell. Across a series of different larval families produced by controlled crosses of pedigreed lines, the effect of experimental ocean acidification ranged from no effect (resilience) to a 28% decrease in shell size. This insight shows, importantly, that genetic-based resilience to climate change may exist and that, moving forward, breeding programs could provide a potential long-term solution for commercial aquaculture.
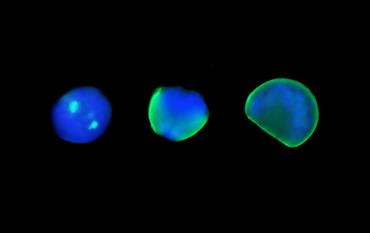
Sequence of first-shell formation in Pacific oyster. The image depicts the overlay of two fluorescence channels to distinguish cell nuclei (blue-stained DNA) from calcified shell (green) across three larval stages. On the far left is a trochophore larva (13-hours post-fertilization), showing localization of first calcification for each valve as two bright green regions. In the middle is a larva that is about half-way through extending its shell around the larval body (16-hours post-fertilization). On the far right is a 1-day-old veliger larva that has a fully developed shell of ~70 µm in length that now encompasses the entire larval body.
ICES Journal of Marine Science, Editor’s Choice feature: http://ices.dk/news-and-events/news-archive/news/Pages/IJMS-Editor%E2%80%99s-Choice-%E2%80%93-the-metabolic-cost-of-ocean-acidification.aspx